[임상연구 동향] 글로벌 줄기세포 임상 현황
한국줄기세포학회
view : 2896
- 검색조건 (stem cells | Recruiting, Not yet recruiting, Active, not recruiting, Completed, Enrolling by invitation,
Suspended, Terminated Studies | Phase Early Phase 1, 1, 2, 3)
- Clinicaltrials.gov에서 등록된 줄기세포 치료 임상연구(Early Phase 1~ Phase 3) 중 중도포기와 시험단계가 알려지지 않은 연구를 제외하고 총 5,049개의 연구가 등록되어 있습니다.
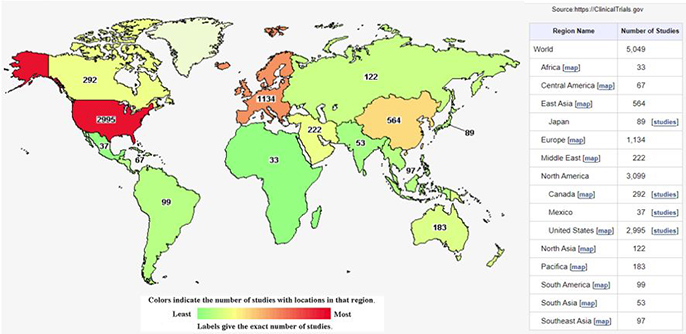
[그림1. 글로벌 줄기세포 임상시험 현황]
- 검색조건 (stem cells | Recruiting, Not yet recruiting, Active, not recruiting, Completed, Enrolling by invitation,
Suspended, Terminated Studies | Phase Early Phase 1, 1, 2, 3| Start date on or after 01/01/2020)
- 이중 최근 2년인 2020년 1월 1일 이후로 현재까지 clinicaltrials.gov에 줄기세포를 활용한 등록된 임상연구는 818건으로 확인되었습니다.
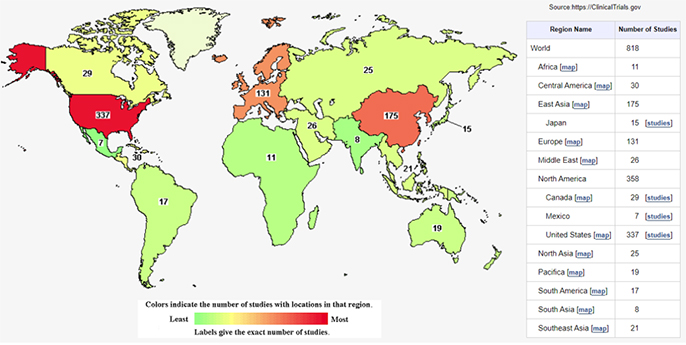
[그림2. 2020년 이후 글로벌 줄기세포 임상시험 현황]
최근 2년간 수행되고 있는 임상시험의 조건 혹은 질환에 대해 항목별로 살펴본 결과 22개의 조건 혹은 질환에 대한 연구가 수행되고 있었습니다.
[그림3. 2020년 이후 글로벌 줄기세포의 질환 혹은 진단]
이러한 줄기세포 치료제 개발을 목표로 수행되는 임상시험에 사용되는 줄기세포는 종류는 다음과 같이 다양한 줄기세포를 활용되고 있음을 알 수있습니다.
| 치료제로 활용되는 줄기세포 |
| Expanded autologous bone marrow-derived mesenchymal stem cell |
| Mesenchymal Stem Cells |
| Autologous Non-Hematopoietic Peripheral Blood Stem Cells (NHPBSC) |
| Olfactory mucosa-derived mesenchymal stem cells |
| Umbilical Cord Derived Mesenchymal Stem Cell (UC -MSC) Transplantation |
| Allogeneic Umbilical Cord Mesenchymal Stem Cell |
| Wharton Jelly Mesenchymal stem cells |
| Pooled mesenchymal stem cell |
| Neural Stem Cells |
| adipose-derived mesenchymal stem cells |
| limbal stem cells |
마지막으로 818개의 임상을 모두 제공하기 어려워 그중 검색해서 제시된 상단 20개를 정리하였습니다.
| NCT Number | Study Title | Status | Interventions | Sponsor/ Collaborators | Phases | Funded Bys | Study Start | Locations |
|---|---|---|---|---|---|---|---|---|
| NCT04519671 | Mesenchymal Stem Cells for the Treatment of Perianal Fistulizing Crohn's Disease | Recruiting | Drug: Mesenchymal Stem Cells Other: Placebo | Amy Lightner The Cleveland Clinic | Phase 1 Phase 2 | Other | 19-Nov-20 | USA |
| NCT04519697 | Mesenchymal Stem Cells for the Treatment of Rectovaginal Fistulas in Participants With Crohn's Disease | Recruiting | Drug: Mesenchymal Stem Cells Other: Placebo | Amy Lightner The Cleveland Clinic | Phase 1 Phase 2 | Other | 28-Oct-20 | USA |
| NCT04473170 | Study Evaluating the Safety and Efficacy of Autologous Non-Hematopoietic Peripheral Blood Stem Cells in COVID-19 | Completed | Biological: Autologous Non-Hematopoietic Peripheral Blood Stem Cells (NHPBSC) Drug: COVID-19 standard care | Abu Dhabi Stem Cells Center Abu Dhabi Stem Cells Center | Phase 1 Phase 2 | Other | 04-Apr-20 | UAE |
| NCT04519684 | Study of Mesenchymal Stem Cells for the Treatment of Ileal Pouch Fistula's in Participants With Crohn's Disease | Recruiting | Drug: Mesenchymal stem cells Other: Placebo | Amy Lightner The Cleveland Clinic | Phase 1 Phase 2 | Other | 28-Oct-20 | USA |
| NCT04776239 | Allogeneic Mesenchymal Human Stem Cell Infusion Therapy for Endothelial DySfunctiOn in Diabetic Subjects With Symptomatic Ischemic Heart Disease. (ACESO-IHD) | Recruiting | Drug: 100 million Allogeneic Mesenchymal Human Stem Cells Other: Placebo | Joshua M Hare National Heart, Lung, and Blood Institute (NHLBI) University of Miami | Phase 1 Phase 2 | Other / NIH | 16-Aug-21 | USA |
| NCT04441658 | Efficacy and Safety of Umbilical Cord Mesenchymal Stem Cells Transplantation in Patients With Type 2 Diabetes Mellitus | Recruiting | Biological: Umbilical Cord Mesenchymal Stem Cells Biological: saline | Shanghai East Hospital | Phase 1 Phase 2 | Other | 10-Apr-20 | China |
| NCT04446897 | Treatment of Chronic Medium-severe Periodontitis With Mesenchymal Stem Cells Predifferentiated in Osteogenic Direction | Completed |
Biological: mesenchymal stem cells Other: standard treatment |
Institute of Biophysics and Cell Engineering Institute of Biophysics and Cell Engineering of National Academy of Sciences of Belarus Belarusian Medical Academy of Post-Graduate Education |
Phase 1 Phase 2 |
Other | 03-Aug-20 | Belarus |
| NCT04972890 | The Outcomes of Intracavernosal Umbilical Cord Mesenchymal Stem Cells Implantation in Patients With Diabetic Erectile Dysfunction | Recruiting | Biological: stem cells Other: placebo | Indonesia University | Phase 2 Phase 3 | Other | 27-Oct-20 | Indonesia |
| NCT04194671 | Clinical Trial of Mesenchymal Stem Cells in the Treatment of Severe Acute Kidney Injury | Not yet recruiting | Biological: Mesenchymal stem cells Other: Saline | Chinese PLA General Hospital | Phase 1 Phase 2 | Other | 31-Dec-21 | China |
| NCT04125329 | Umbilical Cord Mesenchymal Stem Cells Therapy for Diabetic Nephropathy | Recruiting | Drug: Human umbilical cord mesenchymal stem cells | Yan'an Affiliated Hospital of Kunming Medical University | Early Phase 1 | Other | 01-Apr-20 | China |
| NCT Number | Study Title | Status | Interventions | Sponsor/ Collaborators | Phases | Funded Bys | Study Start | Locations |
| NCT05019287 | Menstrual Blood Stem Cells in Severe Covid-19 | Completed | Biological: Allogeneic human menstrual blood stem cells secretome Other: Intravenous saline injection | Avicenna Research Institute Tehran University of Medical Sciences | Phase 1 Phase 2 | Other | 17-Apr-21 | Iran |
| NCT03184935 | Research for Human Umbilical Cord Mesenchymal Stem Cells in the Treatment of Myelodysplastic Syndrome (MDS) | Suspended | Biological: Allogeneic umbilical cord mesenchymal stem cells Drug: Decitabine | Sclnow Biotechnology Co., Ltd. | Phase 1 Phase 2 | Industry | Oct-21 | China |
| NCT04314687 | Stem Cell and Conditioned Medium for Cerebral Palsy | Recruiting | Biological: Umbilical Cord Mesenchymal Stem Cells Biological: Conditioned Medium Other: Standard Therapy |
PT. Prodia Stem Cell Indonesia PT. Prodia Stem Cell Indonesia |
Phase 1 Phase 2 | Industry | 25-Jun-20 | Indonesia |
| NCT03180463 | The Study of Early Stage Osteonecrosis of Femoral Head With Human Umbilical Cord Mesenchymal Stem Cells | Suspended | Drug: Allogeneic umbilical cord mesenchymal stem cells (SCLnow 19#) Procedure: core decompression | Sclnow Biotechnology Co., Ltd. | Phase 1 Phase 2 | Industry | Oct-21 | China |
| NCT05075811 | Study of Ossium Mesenchymal Stem Cells for the Treatment of Pouch Fistulas in the Setting of Crohn's Disease | Not yet recruiting | Drug: Ossium vBM-MSC Other: Placebo | Amy Lightner Ossium Health, Inc. The Cleveland Clinic | Phase 1 Phase 2 | Other / Industry | 15-Nov-21 | USA |
| NCT05008588 | Combination of Conditioned Medium and Umbilical Cord-Mesenchymal Stem Cells Therapy for Sub-Acute Stroke Infarct | Not yet recruiting | Biological: Conditioned Medium Biological: Umbilical Cord Mesenchymal Stem Cells Procedure: Neurologic and Neutrophic Drugs |
PT. Prodia Stem Cell Indonesia PT. Prodia Stem Cell Indonesia |
Phase 1 Phase 2 | Industry | Sep-21 | Indonesia |
| NCT04432467 | Fertility Restoration Using Autologous Mesenchymal Stem Cells | Completed | Biological: Autologous mesenchymal stem cells Other: standard treatment | Institute of Biophysics and Cell Engineering of National Academy of Sciences of Belarus Belarusian Medical Academy of Post-Graduate Education | Phase 1 Phase 2 | Other | 01-Oct-20 | Belarus |
| NCT04631406 | A Safety and Tolerability Study of Neural Stem Cells (NR1) in Subjects With Chronic Ischemic Subcortical Stroke (ISS) | Recruiting | Biological: Neural Stem Cells | Gary Steinberg California Institute for Regenerative Medicine (CIRM) Stanford University | Phase 1 Phase 2 | Other | 04-Jan-21 | USA |
| NCT04651855 | The Evaluation of the Effect of Mesenchymal Stem Cells on the Immune System of Patients With ALS | Recruiting | Drug: Mesenchymal stem cells isolated from Wharton's jelly |
Polski Bank Komorek Macierzystych JSC (PBKM) National Center for Research and Development, Poland |
Phase 1 Phase 2 | Industry / Other | 02-Dec-20 | Poland |
| NCT04661644 | Efficacy and Safety of Mesenchymal Stem Cell Clusters in Patients With Critical Limb Ischemia | Recruiting | Biological: Clusters of adipose-derived mesenchymal stem cells (Dose: 1 x 10^7 cells/1 mL/vial) Biological: Clusters of adipose-derived mesenchymal stem cells (Dose: 1 x 10^8 cells/1 mL/vial) | S.Biomedics Co., Ltd. Dt&Sanomedics | Phase 1 Phase 2 | Industry | 04-Nov-20 | Korea |
